With concerns growing over the burden Leqembi could impose on healthcare infrastructure, Eisai and Biogen have trotted out data for an under-the-skin version of the Alzheimer’s disease drug that may ease those concerns.
An injected version of Leqembi worked as well as the FDA-approved intravenous version at removing the toxic amyloid protein implicated in Alzheimer’s, according to data presented at the Clinical Trials on Alzheimer’s Disease annual meeting.
The data came from an extension of the phase 3 Clarity AD trial, which won Leqembi its FDA approval. Compared with the original version, a subcutaneous injection of the drug cleared 14% more amyloid plaque in the brain among 71 patients who hadn’t received Leqembi before.
The biomarker analysis, done after six months of treatment, was preliminary. There was no data comparing the two formulations on their ability to slow cognitive decline.
Despite the positive efficacy data, a closely watched safety measurement raised some eyebrows. But analysts argued the readout shouldn’t be a concern.
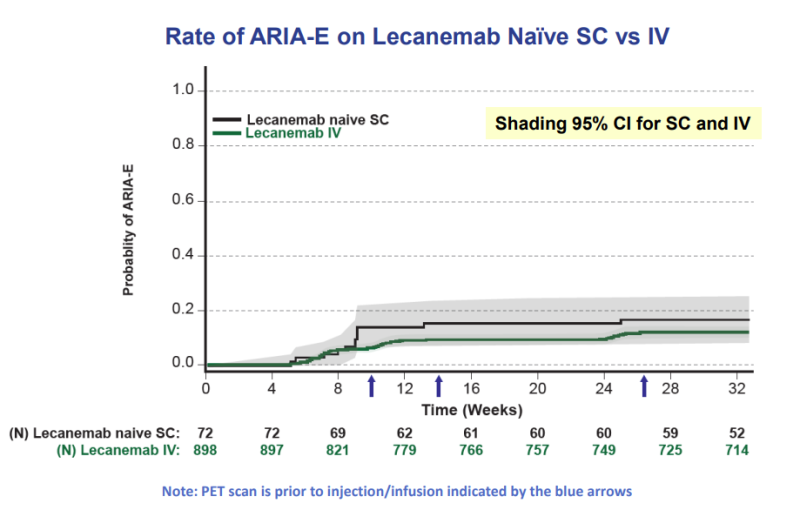
Specifically, subcutaneous Leqembi showed a numerically higher rate of amyloid-related imaging abnormalities (ARIA), a known side effect of anti-amyloid antibodies like Leqembi. The rates of brain swelling, known as ARIA-E, were 16.7% and 12.6% for subcutaneous (SC) and intravenous (IV) Leqembi, respectively. The rates of small brain bleedings, known as ARIA-H, were 22.2% and 17.3%, respectively.
ARIA is a potentially life-threatening condition. The side effect has been a major concern when evaluating the benefit-risk profile and commercial potential of Leqembi and other anti-amyloid drugs.
But analysts from Mizuho, Leerink Partners and William Blair all separately pointed out that the ARIA difference was drawn from a very small number of patients. The safety analysis included 72 patients on SC and 898 on IV. One patient in the SC group included in the safety analysis discontinued the trial for reasons not related to the medication and was not part of the efficacy analysis, an Eisai spokesperson told Fierce Pharma.
Because of the small number of patients, saying SC is worse than IV on ARIA-E is “splitting hairs” and simply “not true,” Mizuho analyst Salim Syed said in his note Thursday.
Syed called the two versions’ ARIA profiles “more similar than dissimilar.” In a statistical analysis that mapped out the range where the true ARIA-E rates could fall, the SC and IV versions overlapped with each other.
Still, the seemingly higher ARIA rate was not the best news for Eisai. As a team of William Blair analyst noted, Eisai had recently predicted that the SC dosing could potentially lead to lower ARIA incidence given that it has lower peak concentration in the bloodstream.
But based on the current study, it appears that the steady-state exposure—rather than maximum exposure in IV—is a better predictor of ARIA-E rates in the subcutaneous version, the Eisai spokesperson explained. Eisai and Biogen are proposing self-administered Leqembi as a weekly injection, which offers a more stable exposure to the drug than the biweekly IV formulation.
As Syed and the William Blair team see things, SC Leqembi’s ARIA data look better than what Eli Lilly has shown for its rival candidate donanemab, which is expecting an FDA decision by the end of this year.
For their part, Eisai and Biogen are preparing to file an FDA application for Leqembi’s SC version in the first quarter of 2024. Overall, analysts at Leerink called the latest SC data “good news” for the two companies.
The SC version could offer another dosing option with a comparable efficacy-safety profile and could enable more convenient at-home dosing, the Leerink analysts said. The results bode well for Leqembi’s commercial opportunity, the team added.
The large size of the Alzheimer’s patient population has prompted concerns that the existing healthcare infrastructure at hospitals and infusion centers won't be able to handle the diagnosis, administration and monitoring required by Leqembi and other anti-amyloid drugs.
Mizuho’s Syed projected that Leqembi could reach $12 billion in peak sales, even after factoring in a “fair amount” of competition from Lilly’s donanemab. The William Blair team put Leqembi’s 2030 global revenue at $8 billion.