After publishing promising preclinical results, a team from the Scripps Research Institute, led by researcher William Schief, are taking their HIV vaccine candidate to federal regulators to discuss human trials.
Using the engineered antigen eOD-GT8, the team showed that their candidate generated the antibodies necessary to jumpstart an initial immune reaction against the virus types contained in HIV.
The group sought to create broadly neutralizing antibodies--which can eliminate a vast range of virus types--to combat the collection of virus variants of which HIV consists. In their study, the group found in mice that the candidate has the potential to do just that.
In a separate study published last week, Weill Cornell Medical College's John Moore and colleagues used an engineered trimer called BG505 SOSIP.664 to generate potent neutralizing antibodies, though with a narrow range of targets. Despite the narrow range, John Mascola, director of the National Institute of Allergy and Infectious Diseases' Vaccine Research Center, told The Scientist the work represents a "major advance" in HIV research and that much progress will be made in the coming months and year building on the platform. NIAID funded the research.
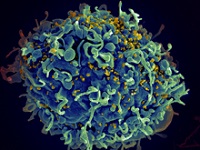 |
| HIV particles infecting a human T cell--Courtesy of NIH/NIAID |
Though their shot isn't expected to prevent HIV infection, Schief's team says their candidate is ready for clinical trials that will let them know if they're on the right track. If patients experience the initial immune reaction, then the team can work to formulate immunogens to create the antibodies necessary to combat HIV.
"We think it's the right time to find that out," Schief told The Scientist, with Mascola agreeing. Mascola added that "both of these approaches have crossed a threshold in the laboratory ... to provide enough scientific rationale for human testing."
Meanwhile, France-based Biosantech reported last week that its HIV vaccine candidate was not toxic for patients in a study involving 48 HIV-positive patients.
And in April, Duke University scored a $20 million NIH grant to develop its own vaccine, while San Diego-based Vical ($VICL) in the same month inked a $4 million contract with the IPPOX Foundation to supply plasmid DNA for HIV vaccine trials, funded by a grant from the Bill and Melinda Gates Foundation.
- more from The Scientist