Tokyo, Japan, April 10, 2015 – Daiichi Sankyo Co., Ltd. (hereafter, Daiichi Sankyo) and Terumo Corporation (hereafter, Terumo) today announced that Japan Vaccine Co., Ltd. (hereafter, Japan Vaccine), an affiliated company of Daiichi Sankyo, has filed an application for the manufacture and sales approval in Japan for an intradermal seasonal influenza vaccine (hereafter, The Vaccine).
The Vaccine is a pre-filled syringe type, intradermal influenza HA vaccine*1 co-developed by four companies [Daiichi Sankyo, Terumo, Japan Vaccine and Kitasato Daiichi Sankyo Vaccine Co., Ltd (hereafter, Kitasato Daiichi Sankyo)] under a February 23, 2012, agreement between Daiichi Sankyo and Terumo for the co-development of an intradermal vaccine for infectious diseases.
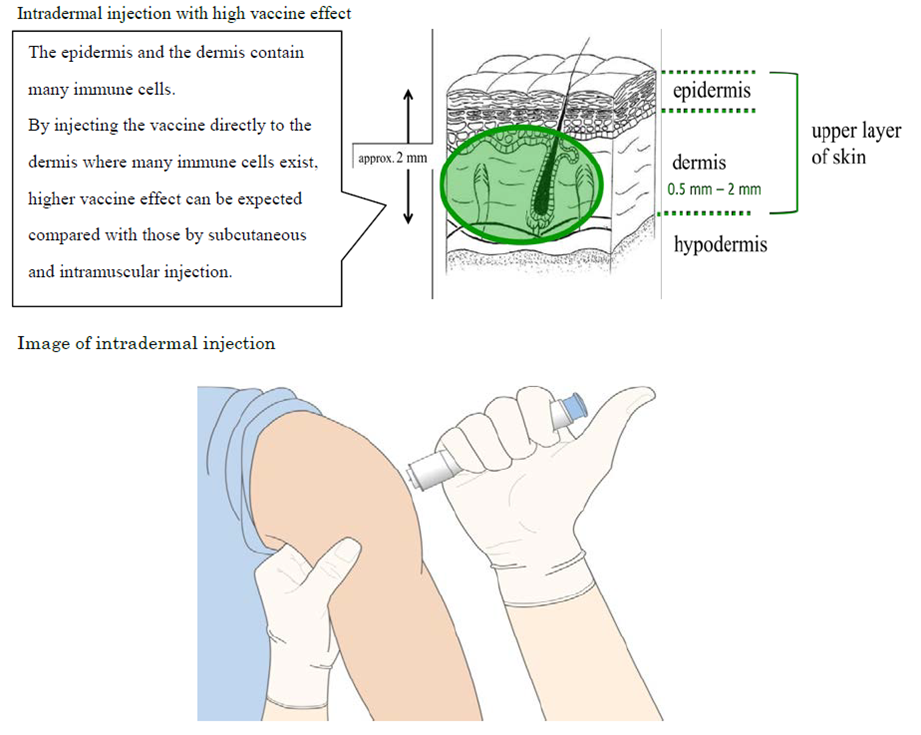
Terumo developed the intradermal injection*2 device, which offers a more easy-to-use, surefire method to administer the vaccine than current methods. The device is also expected to ease patient hesitation to be injected and lower the risk of damaging peripheral blood vessels and nerves within the subcutaneous tissue.
Daiichi Sankyo is committed to contributing to further the prevention of seasonal influenza through The Vaccine, as well as contributing to the protection of people's health by working to promote and enrich the preventive treatment environment and improve hygiene through the production of practical vaccines which meet medical demands.
Terumo aims to provide a new value to the medical front by combining high specialties in both pharmaceutical products and medical devices. Through these innovative developments, Terumo aims to establish itself as a company with a global presence in the medium to long-term.
*1 An intradermal injection device developed by Terumo, which is a combination of a pre-fillable syringe with a needle specially designed for intradermal injection, pre-filled with influenza HA vaccine developed by Kitasato Daiichi Sankyo.
*2 Recent study reports improved antigen-recognition by the immune system and enhanced effect of the vaccine through intradermal vaccine injection.
(Hickling JK, Jones KR, Friede M, Zehrung D, Chen D, Kristensen D: Intradermal delivery of vaccines: potential benefits and current challenges. Bull World Health Organ 89: 221-226, 2011.)