 |
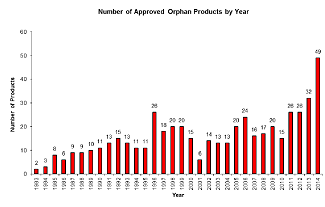
| Orphan drug approvals climbed 53% to 49 in 2014. --Courtesy FDA Law Blog (Click for more info) |
New drug approvals were up last year, and orphan drug approvals were no exception. But by just how much did they climb?
The FDA Law Blog has done some digging through the agency's Orphan Drug Designations and Approvals database, and it's got an answer. 2014 saw 49 orphan drug approvals--a 53% increase over 2013. Those approvals aren't showing any signs of slowing down, either, and neither are pharma companies: Right now, orphan drug designation requests are on pace to break last year's record.
It's a good sign for drugmakers on multiple fronts, the blog notes. For one, other countries are looking at creating similar programs, which could be good for approval stats overseas. But copycats are popping up on the FDA's home turf, too; take the Generating Antibiotic Incentives Now Act, for example, which also incentivizes drugmakers to focus their efforts on a comparatively neglected field.
The more efforts there are out there to speed along drug production, the faster drugmakers will be seeing sales. And considering the often sky-high price tags on orphan drugs, even a little more time on the market can make a big difference.
It's those same sky-high price tags that may pose hurdles for orphan drugmakers going forward, however. Traditionally, they've managed to defend their high prices with their small patient pools. But with non-orphan drugmakers stirring the pot with their own lofty stickers--like Gilead, whose $84,000-per-12-week-course hep C med Sovaldi first began drawing fire in late 2013--penny-pinching payers are on their guard.
Orphan drugmakers know as much, though, and some are trying to manage the situation. Take Vertex ($VRTX), whose Kalydeco currently boasts approval for a fraction of cystic fibrosis sufferers. Company execs recently said they're taking steps to make sure they can keep prices where they are when and if the drug wins broader approval--without touching off a firestorm.
With the drug expected to win a major label expansion later this year that could bring its eligible patient headcount from 1,650 to 8,500 in the U.S., Chief Commercial Officer Stuart Arbuckle seemed to be preparing payers for the financial blow on Vertex' Q4 conference call last month. The company is "investing in disease education to help payers estimate the number of eligible patients they may have in their plans," he told investors.
- get more from the FDA Law Blog
Special Reports: Top 20 orphan drugs by 2018 | Biopharma posts a chart-topping 41 new drug approvals in 2014