A far-reaching indictment against 14 New England Compounding Center (NECC) owners and employees not only accuses them of peddling unsterile drugs that infected hundreds of patients with fungal meningitis, but also includes 25 murder charges against two lead pharmacists, a move that will not go unnoticed throughout the entire pharma industry.
The 131-count criminal indictment, unsealed in Boston Wednesday in connection with the 2012 nationwide fungal meningitis outbreak, charges owner and head pharmacist Barry J. Cadden and NECC supervisory pharmacist Glenn A. Chin with 25 acts of second-degree murder for deaths of patients in 7 states. If convicted, they face up to life in prison. Chin, who was intercepted by federal agents when he was about to board a plane for Hong Kong last September, has already pleaded not guilty to some of the charges, according to Bloomberg.
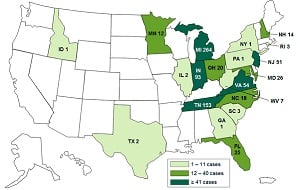 |
| A CDC case count map for the 2012 fungal meningitis outbreak (Click for a larger version) |
"Senior N.E.C.C. pharmacists knew that, despite the filthy conditions at N.E.C.C., the drugs that they made were not properly tested for sterility," Carmen Ortiz, the United States attorney for Massachusetts told The New York Times.
The 73-page document alleges the now defunct New England Compounding Center reaped millions of dollars by cutting corners in the manufacture of what were supposed to be sterile drugs, falsifying records, using expired ingredients and lying to clients about their safety. The result was that drugs became contaminated with fungal meningitis infecting 751 people and killing 64 people who had received injections of a steroid for pain.
While murder charges are usually a state offense, The New York Times points out that in cases of racketeering, federal murder charges can be tacked on. The U.S. Attorneys office said in a statement to the newspaper that prosecutors do not have to prove intent to get murder convictions against Cadden and Chin, only that they "acted with extreme indifference to human life."
The prosecutor's office in Boston was unable to speak to what precedence, if any, this poses for the larger pharmaceutical industry but Columbia University law professor Daniel C. Richman noted the seriousness to the Times. "It certainly is a dramatic move to label what was happening as fraud and tie that to murder, instead of casting it as regulatory violations," Richman said. "That frames the misconduct in stark criminal terms."
The allegations were laid out in a 73-page indictment handed up by a federal grand jury Wednesday in Massachusetts, the Department of Justice announced Wednesday. Among the allegations is that NECC often cut short the autoclaving of drugs, slighted sterility testing and when tests did find microbial growth, the company did not recall suspect drugs.
Eleven of the 14 were arrested Wednesday, Bloomberg reported. Among those indicted are Gene Svirskiy, a supervising pharmacist in one of the "clean rooms" and Robert Ronzio, the company's national sales director. Also indicted were Greg, Douglas and Carla Conigliaro, shareholders of the company. The Conigliaro family of Massachusetts opened NECC in 1998, the Times reports, as one business in a large organization that includes a recycling firm. Carla and Douglas Conigliaro, who are married, are accused of transferring $33 million in assets to 8 different bank accounts after the pharmacy went into bankruptcy and a court ordered all assets frozen. The couple pleaded not guilty, the Times reports and their lawyer claimed the transfers were all legal.
 |
| FDA Commissioner Margaret Hamburg |
The 2012 outbreak brought stark attention to the growth of the drug compounding industry, where large operations are mass producing drugs and using sales forces to market them nationally. FDA Commissioner Margaret Hamburg faced tough scrutiny from Congress about why the FDA was not inspecting most compounding facilities. The public outcry over the episode led to Congress passing the Drug Quality and Security Act, which gives the FDA new, but limited, powers to oversee compounding pharmacies that volunteer to be regulated.
While it did not get the authority it sought, the agency has the right to inspect compounding facilities when there are questions of safety and it has inspected dozens of them, requiring many to recall all of their drugs that the FDA believed were manufactured under questionable conditions. This month the FDA elicited a guilty plea to a federal criminal misdemeanor from the co-owner of Main Street Family Pharmacy, a Tennessee-based compounding pharmacy. The case was tied to its sales of a contaminated steroid that caused 26 people to become ill.
NECC was shut down in October 2012 after authorities traced the outbreak to its facility that produced a tainted steroid, which was distributed to health providers in 20 states. It declared bankruptcy two months later. Earlier this month, a new plan was filed with a bankruptcy court to establish a fund of at least $135 million in compensation for victims and families affected by the outbreak.
- here's the DOJ release
- here's the indictment
- read the Bloomberg story
- more from the New York Times