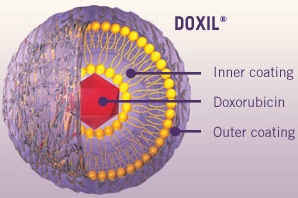
 |
| The FDA approved Janssen's process of manufacturing bulk Doxil at an Ohio facility.--Courtesy of J&J |
In December, Janssen signed a 6-month lease to take over the portion of a Ben Venue plant in Ohio that had manufactured cancer med Doxil for the Johnson & Johnson ($JNJ) unit. Ben Venue was closing the plant, and Janssen was scrambling for a short-term solution to maintain supplies of the ovarian cancer drug now that its contractor was closing up shop. The FDA has now signed off on Janssen's process of manufacturing bulk Doxil at the Ohio facility. It is part of an "alternative manufacturing process," in which it hands off fill and finish of the drug to another manufacturer.
Janssen spokeswoman Lisa Vaga said Monday that "the FDA has accepted our interim control plans at the Bedford facility to handle the bulk manufacturing for the period of the lease agreement. Under the lease, we plan to continue to release Doxil through FDA regulatory discretion as supplies become available, using this alternate manufacturing approach." That is the near-term solution, Vaga said, and in the long term, Janssen is making progress toward agreements with other manufacturers to produce Doxil, also marketed as Caelyx in some countries.
While the FDA has not approved the two-facility process for making Doxil, it has agreed to allow Janssen to release a new lot of the cancer drug made through that process. The company posted a letter to an update site Monday telling healthcare providers that the lot had passed a full Janssen review to ensure that it hit all of the drugmaker's quality and safety standards and would now be available.
Shortages of Doxil first arose in 2011 when Ben Venue, a division of Germany's Boehringer Ingelheim, had to temporarily close the Bedford plant to make upgrades after FDA inspectors found huge issues with it. Janssen's long-term plan was to get new contractors approved to produce the drug. But it needed a short-term solution. In January 2013, Janssen worked out a deal in which the plant made bulk Doxil, which was then shipped to another facility which handled sterile filtration and packaging, Vaga explained in an interview Monday. But the situation got even more complex when Boehringer Ingelheim determined last year that the operating losses it faced at the plant were more than it was willing to absorb and decided to close the plant at the end of the year.
Janssen, which is in litigation with Ben Venue for not living up to its contract to manufacture the drug, extracted an agreement from the Boehringer Ingelheim unit to lease the portion of the plant that had made Doxil. It then sought FDA approval to allow it to step in and handle the bulk product manufacturing and to continue to hand off fill and finish to the contractor.
- here's Janssen's provider letter (PDF)
- here's access to all of the updates