First it was Italy, then it was France, and now the European Union is stepping up to take a serious look at how Swiss drugmakers Roche ($RHHBY) and Novartis ($NVS) have handled the marketing of their eye drug Lucentis. The EU's antitrust chief, Joaquin Almunia, told Bloomberg that regulators are "gathering information" on whether the two companies colluded to prevent the use of Avastin--a much cheaper drug that's similar to Lucentis--for treating age-related macular degeneration (AMD).
In early March, Italy's antitrust authority levied more than $250 million in fines against Roche and Novartis, on charges that the companies catalyzed sales of Lucentis and Avastin by pushing doctors toward the pricier option. The companies continue to deny the charges, but soon after the fines were imposed, Italian prosecutors began investigating whether they engaged in market manipulation and fraud.
By mid-March, France was in on the probe, going public with the revelation that it actually has been investigating the Lucentis-Avastin controversy since 2012, when French Health Minister Marisol Touraine first expressed concerns about possible collusion. On April 10, the French Competition Authority raided local offices of Roche and Novartis looking for evidence of collusion.
 |
| EU antitrust chief Joaquin Almunia--Courtesy of Agencia Brasil, CC BY 3.0 BR |
Almunia isn't saying much yet about the EU's investigation beyond acknowledging that it involves close collaboration with French and other European authorities, according to Bloomberg. A spokesman for Roche told Bloomberg there is no agreement between it and Novartis to restrict competition.
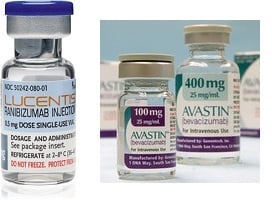
Avastin, which is approved to treat some cancers, and Lucentis have the same mechanism of action--they both choke off blood vessels--but Roche's California unit, Genentech, developed Lucentis specifically for ophthalmic use. Lucentis costs about $2,000 per dose, while Avastin sells for just $100 per dose, prompting ophthalmologists all over the world to repackage the cheaper drug for use in patients with AMD.
The EU's case was partly prompted by an advocacy group called European Consumer Organisation (known as BEUC), which has been lobbying heavily for an investigation, according to Bloomberg. "This case undoubtedly requires an EU-wide investigation as both medicines have been approved" in the EU, Ilaria Passarani, senior health policy officer at BEUC, told Bloomberg. "Consumers have the right to access cheaper medicines."
-here's the Bloomberg story