 |
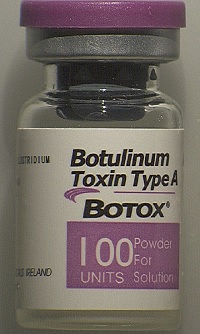
| Counterfeit Botox vial--Courtesy of the FDA |
More counterfeit Botox has made its way into the U.S. and, the FDA thinks, into physician offices. The agency is warning providers that they need to make sure they don't have any of it because it is believed to be unsafe.
The product was sold by an unauthorized supplier and there is no way of knowing if the product was maintained at proper temperatures during storage and shipment, the agency said in a website posting. Botox is a sterile, purified version of the same toxin that causes botulism and must be stored and distributed following exacting standards.
While the packaging is close in appearance to the original from Allergan ($AGN), the FDA says there are some telltale signs to look for. The vial is missing the lot number while the outer carton does not have any entries next to the LOT: MFG: EXP, the FDA said. Also, the outer carton and vial display the active ingredient as "Botulinum Toxin Type A" instead of "OnabotulinumtoxinA."
The FDA has been through this drill before. In 2012 it warned 350 doctor practices that supplies of Botox they bought through companies with ties to Internet pharmacy CanadaDrugs.com were at best unapproved foreign versions--and therefore had been sold to them illegally--and could in fact be counterfeit. Months later, the FDA and Allergan, now owned by Actavis ($ACT), warned providers that a foreign version of the drug had been discovered in counterfeit packaging that made it look like it was the U.S. version.
In that case, the products were sold with names like "Onlinebotox" and were being offered on the cheap through "blast faxes." Again, because there was no way of knowing anything about the supply chain process, there was no way to know if it was safe to be injected into patients' faces. Besides being approved to smooth out wrinkles, it is also approved for treating headaches, overactive bladders and other conditions.
One tactic of U.S. authorities in fighting the onslaught of illegally imported and sometimes counterfeit drugs is to prosecute doctors who bought them knowing they came from outside the legitimate supply chain. In 2013, a 41-year-old Missouri doctor was given a 5-month prison sentence for lying to federal agents about buying a foreign-made version of the drug online. He told them he had bought it three times, when in fact, he had made four dozen purchases from an online seller whose contact was a Gmail account.
- here's the FDA warning