New CE mark in the European Union extends application to routine pathology including primary diagnosis with human tissue specimens
TUCSON, Ariz., Aug. 20, 2014 /PRNewswire/ -- Ventana Medical Systems, Inc. (Ventana), a member of the Roche Group, today announced that its VENTANA System for Primary Diagnosis1 has been CE marked in the European Union for routine pathology, including primary diagnosis with human tissue specimens. The system, consisting of VENTANA Virtuoso software coupled with either the VENTANA iScan Coreo or the VENTANA iScan HT slide scanner, provides automated digital slide creation, case management and computer viewing of human tissue specimens. This new CE mark for their application in routine pathology including primary diagnosis takes digital pathology a big step forward by expanding diagnostic options for a lab's most common test—the Hematoxylin and Eosin (H&E)—while improving workflow, physician collaboration and the standard of patient care.
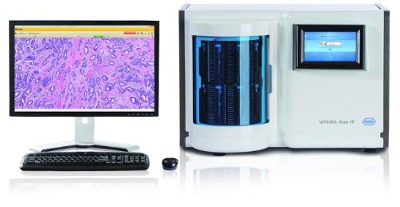
"As more anatomic pathology laboratories routinely digitize patient slides, it's important that the most critical and commonly prepared stain—the H&E—can be reliably scanned and interpreted by a pathologist using a computer monitor for primary diagnostic purposes," said Dr. Steve Burnell, PhD, VP and Lifecycle Leader for Digital Pathology and Workflow at Ventana. "This CE marking under the European Union's In Vitro Diagnostics Directive (IVDD) represents another significant step by Roche to assist pathologists in transitioning to a digital workflow and providing the highest standards of patient care."
Digital pathology adoption is accelerating as healthcare organizations face regional imbalances of pathology expertise. Digital pathology solutions offered by Ventana continue to improve patient care by enabling pathologists worldwide to gain access to physician networks offering sub-specialty pathology expertise.
The iScan Coreo slide scanner is a brightfield 160-slide capacity scanner that provides a versatile system including four objective lenses as well as live mode viewing. The VENTANA iScan HT slide scanner is a high-throughput brightfield slide scanner with a 360-slide capacity, combining high reliability and high throughput at both 20X and 40X magnifications. Slides digitized on either scanner may be viewed and diagnosed in the Virtuoso web-based software solution for viewing, managing and sharing digital cases and glass slides.
In addition to the VENTANA System for Primary Diagnosis, Ventana recently announced the FDA clearance for its Estrogen Receptor (ER) Image Analysis and Digital Read Application for breast cancer, making it the only company in the industry today offering an FDA-cleared testing package of five key breast biomarkers with their corresponding image analysis algorithms and digital read applications for cancer patients. All IHC breast markers in the Ventana portfolio have both image analysis and digital read application FDA 510(k) and CE IVD clearances. Along with the Companion Algorithm ER (SP1)2 image analysis software, the full breast panel includes HER2 (4B5), PR (1E2), Ki-67 (30-9) and p53 (DO-7) image analysis algorithms along with their accompanying VENTANA IHC assays3.
1The VENTANA System for Primary Diagnosis has not been approved or cleared by the FDA for use in the US.
2When the VENTANA ER (SP1) algorithm is used in conjunction with the CONFIRM anti-ER (SP1) Rabbit Monoclonal Primary Antibody test, it may be used as an aid in the assessment of ER status in breast cancer patients for whom endocrine treatment is being considered but is not the sole basis for treatment.
3The PATHWAY HER2 (4B5) assay is FDA-approved, the CONFIRM PR (1E2) and CONFIRM ER (SP1) assays are FDA-cleared, and Ki-67 (30-9) and p53(DO-7) assays are FDA class 1, exempt in vitro diagnostics.
About Ventana Medical Systems, Inc.
Ventana Medical Systems, Inc. ("VMSI") (SIX: RO, ROG; OTCQX: RHHBY), a member of the Roche Group, innovates and manufactures instruments and reagents that automate tissue processing and slide staining for cancer diagnostics. VENTANA products are used in clinical histology and drug development research laboratories worldwide. The company's intuitive, integrated staining, workflow management platforms, and digital pathology solutions optimize laboratory efficiencies to help reduce errors, support diagnosis and enable informed treatment decisions by anatomic pathology professionals. Together with Roche, VMSI is driving Personalized Healthcare through accelerated drug discovery and the development of "companion diagnostics" to identify the patients most likely to respond favorably to specific therapies. VENTANA, the VENTANA logo, ISCAN, VENTANA ISCAN, VIRTUOSO and COMPANION ALGORITHM are trademarks of Roche.
VMSI Media Relations
Jacqueline Bucher
Senior Director, Corporate Communications
Phone: 520-877-7288
e-mail: Jacquie Bucher
Photo - http://photos.prnewswire.com/prnh/20140819/137678
SOURCE Ventana Medical Systems, Inc.