Has India given up the compulsory license fight? According to a U.S. trade group, officials have privately promised not to grant any more of the licenses, which force branded drugmakers to allow generics companies to knock off their on-patent drugs.
As Reuters reports, the U.S.-India Business Council assured the U.S. Trade Representative that it's no longer open to compulsory license requests from domestic drugmakers. The disclosure came in a USIBC submission to the trade rep, which is working on an annual report about international trade barriers.
Under Indian law--and World Health Organization protocols--the government is allowed to open the door to early generic competition when a medicine is too pricey for local use, but important to public health.
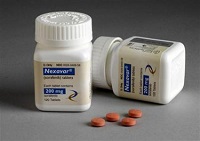 The threat of compulsory licensing became all too real in 2012, when Bayer was forced to allow India-based Natco Pharma to market a copycat version of its branded cancer drug Nexavar. Since then, a raft of domestic pharma companies have filed for compulsory licenses on various meds, but so far, patent officials have not granted any other requests.
The threat of compulsory licensing became all too real in 2012, when Bayer was forced to allow India-based Natco Pharma to market a copycat version of its branded cancer drug Nexavar. Since then, a raft of domestic pharma companies have filed for compulsory licenses on various meds, but so far, patent officials have not granted any other requests.
But compulsory licensing has not been the only way India has blocked Big Pharma in an attempt to keep down drug prices. Patent officials have nullified IP protections on brands sold by Pfizer ($PFE), Roche ($RHHBY), Bristol-Myers Squibb ($BMY) and others. The patent office has also refused to grant patents on drugs to start with, such as a formulation of Novartis' ($NVS) leukemia med Gleevec. Indian courts have been less than sympathetic to multinational drugmakers' appeals in those cases.
In recent months, some official decisions suggested that India was backing away from its patent fights with Big Pharma, perhaps because of intense pressure from the U.S. For instance, in January, India's patent board denied a compulsory license request on Bristol-Myers Squibb's diabetes drug Onglyza. Hyderabad-based Lee Pharma had petitioned for the license.
Yet, as recently as December, Indian officials refused to give up its right to grant compulsory licenses, but said they were drafting legislation to change its intellectual property approach on drugs. As FiercePharmaAsia has reported, the proposal would allow the government to hire hundreds of patent lawyers and researchers to speed the process of reviewing patent applications.
The long delays in application processing have been exacerbated by similar shortage of help in Indian courts trying to deal with patent-infringement cases. Generics makers have been taking advantage of the delays by deliberately infringing on multinational drugmakers' IP rights while applications were pending.
- read the Reuters news