The downward trend continues for FDA action letters--but whether that's actually a good thing for the industry remains to be seen.
In 2015, the OPDP issued just 9 letters, down from 10 the previous year. While the one-year drop is small, a further look back points to an ongoing decline since 2010, when there were 52 action letters sent. In the years between, there's been an annual decline in the number of letters. There were 31 sent in 2011; 28 in 2012 and 24 in 2013.
The 2015 stack contained two warning letters and 7 untitled letters. The warnings went to Duchesnay for its Kim Kardashian Instagram post touting Diclegis with only a link to side effects, and to Valeant ($VRX) subsidiary ECR Pharmaceuticals over sales material for its narcotic cough medicine TussiCaps that omitted risk information, offered an inadequate indication and made unsubstantiated claims. The last one sent in 2015 was the Duchesnay cease and desist on Aug. 7.
When asked about the decreasing number of letters, an FDA spokeswoman noted via email to FiercePharmaMarketing that only looking at the letters doesn't provide a complete picture of OPDP efforts.
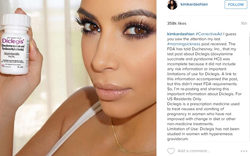 |
| Kim Kardashian issued a corrected social media post for Diclegis in 2015. |
"OPDP uses a risk-based approach to carefully allocate its resources among these activities to have the greatest beneficial public health impact. Therefore, it is apparent that that one cannot get a complete picture of OPDP's program area by looking at a snapshot of time for enforcement letters. Reviewing the number of compliance actions that OPDP takes within a year timeframe does not take into account the work that OPDP does on the other priorities to assist companies with compliance, such as policy and guidance development and core launch reviews, to fulfill its public health mission," she wrote.
Mark Senak, who writes the Eye on FDA blog and has been following the issue for years, declined to guess the reason for the decrease. However, he did say that the lack of letters could be problematic for pharma companies.
"There are two means by which we get the FDA's thoughts on issues. One is guidance documents and the other is the issuance of letters," he said in an interview with FiercePharmaMarketing. "If there aren't more guidance documents and there aren't many letters, it doesn't set up the parameters very well."
Senak also wrote on his blog that it's important especially because of OPDP's "unfinished work" in digital media.
" … Digital media continues to rapidly evolve, bringing new questions into the marketplace. If the agency is silent through the guidance process and silent through the enforcement process then it leaves a large swath of unchartered and unknown territory. That, in turn, leaves a very uneven environment not just for companies, but for patients seeking health information through digital platforms," he wrote.
- read Senak's blog post