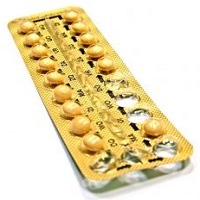
 Four years ago, a unit of Endo Health Solutions recalled 3.2 million blister packs of birth control pills after discovering the pills may have been packaged in the wrong order. Now legal problems tied to the mess-up are swelling for Endo Health Solutions and contractor Patheon as more than 100 women have taken to the courts, saying they got pregnant as a result and want some child support.
Four years ago, a unit of Endo Health Solutions recalled 3.2 million blister packs of birth control pills after discovering the pills may have been packaged in the wrong order. Now legal problems tied to the mess-up are swelling for Endo Health Solutions and contractor Patheon as more than 100 women have taken to the courts, saying they got pregnant as a result and want some child support.
The women have filed suit in a state court in Pennsylvania and are asking the drug companies to pay them millions of dollars, in some cases to cover the costs of giving birth and raising and educating the children that came from the unplanned pregnancies, The Morning Call reports. The U.S. headquarters for Dublin-based Endo are in Malvern, PA. The cases were filed last week after a federal judge earlier this month denied a request for class-action status. Neither Endo nor Patheon would comment.
The publication cites a court order that said that of 117 women from 26 states represented by attorney Keith Bodoh, 113 became pregnant and 94 carried their babies to term. At least one was said to have been given up for adoption by a military mother who was being deployed and one woman dropped out of law school and another out of nursing school after getting pregnant.
The problems started in 2011, when a Kansas City woman returned her birth control pills to the pharmacist after noticing that the blister pack had been rotated 180 degrees, reversing the weekly tablet orientation, The Morning Call reports, citing the lawsuit. With hormonal contraceptives, taking the right pill on the right day is crucial. Qualitest Pharmaceuticals, a unit of Endo, then issued the recall.
Other drugmakers have also been sued over similar packaging missteps. Canadian generics maker Apotex was sued in 2013 by nearly four dozen women after it recalled about half a million packages of contraceptives after discovering some contained 14 active contraceptive pills and 14 placebos instead of the 21 active and 7 placebos that they should contain.
And other companies, including Novartis' ($NVS) Sandoz unit, Pfizer ($PFE), and Glenmark Pharmaceuticals, have all had to recall contraceptives for similar packaging problems. Just last April, Pfizer said that about 100 women in Canada had gotten packages of expired birth control pills after a drug distributor accidentally shipped out the wrong products.
- read the story