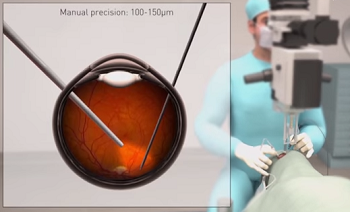
 |
| A depiction of Preceyes' robot-assisted eye surgery--Courtesy of Preceyes |
Two startups--one from the U.K. and another from the Netherlands--have teamed up to combine a robotic device with a gene therapy for high-precision delivery to the subretinal space. It will begin as a development partnership as the companies establish the technique, but the plan is to move into human trials together down the road.
The U.K.'s Nightstar is developing an adeno-associated viral vector-based gene therapy designed specifically for the rare disease choroideremia, a type of hereditary retinal dystrophy for which there is no treatment. Currently, the process involves straightforward eye surgery with an injection and is being tested in 12 patients in an early- to mid-phase trial.
And that's where Preceyes comes in. Preceyes' robotic system offers assistance during eye surgery so that a needle can be monitored, steadied and deployed to an exact spot in the eye.
"The collaborations with Nightstar and the University of Oxford are a major recognition of the unique value of our technology and its applicability to new treatments for unmet needs," Nightstar CEO Gerrit Naus said in a statement. "We are very pleased to work closely with Nightstar and the University of Oxford to further mature the gene-therapy delivery and look forward to market adoption."
A team of scientists at the University of Oxford, led by Robert MacLaren, will be leading human trials using the Preceyes system, though not yet with Nightstar's gene therapy.
"Over the last century, devices that enhance surgical precision have given us the greatest breakthroughs in ophthalmology," MacLaren said in a statement. "We are delighted to have approval for a clinical trial in Oxford which will be the first to assess use of a robotic device for surgery inside the eye."
- here's the release