 |
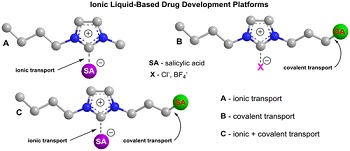
| Representation of three different types of platforms for ionic liquid-based drug development to deliver poorly soluble compounds--Courtesy of Russian Academy of Sciences |
A new way to develop drugs may someday give poorly soluble compounds an edge, overcoming a problem in drug delivery known as polymorphism. A group of scientists from the Russian Academy of Sciences has employed ionic liquids to solubilize those pharmaceutical ingredients that don't dissolve very easily in the body.
Polymorphism is "the ability of solid drugs to form several different crystal structures," according to a release from the Russian Academy, and it sets up some hurdles in the development process. These polymorphs can be so different from each other in terms of properties and performance that they are much less successful as treatments. Also, the process is difficult to control, making the overall drug less efficient in treating a disease.
To deliver the well-known anti-inflammatory compound salicylic acid as a model, the scientists incorporated it into a highly tunable ionic liquid, which gave the drug much higher water solubility (water being a main component of the human body). In treating human fibroblasts and colorectal adenocarcinoma cells, the salicylic acid retained its activity in its new form.
The drug can be connected with the salt (ionic) liquid three different ways: ionic binding, covalent binding and dual binding using both ionic and covalent bonds. With all three, the cytotoxicity was significantly higher, and the third one in particular shows promise as a complex delivery system able to adapt to specific solubility needs with more efficiency.
Down the road, this complex delivery could help deliver dangerous cancer drugs in lower doses because of the higher solubility, cutting down on side effects greatly.
The researchers published their work in the journal ACS Medicinal Chemistry Letters.
- here's the release
- and here's the abstract