 |
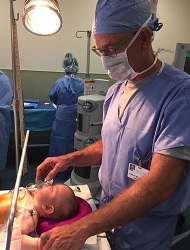
| Anesthesiologist Robert Williams with infant patient--Courtesy of UVM Children's Hospital |
As concerns about general anesthesia for infants and toddlers grow, more and more surgeons are turning to spinal anesthesia methods, which are thought to reduce problems such as memory loss and learning difficulties that have been associated with anesthetic exposure, according to a report from The Wall Street Journal.
Spinal anesthesia isn't a new technique--it's been in use for 40 years, though not very widely. The spinal delivery leaves a baby awake during surgery, and the possibility of movement during the procedure has left many surgeons less willing to adopt the method in the past. But as more studies suggest the possible drawbacks of general anesthesia, surgeons are beginning to make the change.
A large study including 28 hospitals in the U.S., Canada, Europe, Australia and New Zealand, is measuring intelligence in 722 5-year-olds who received either general or spinal anesthesia for hernia repair surgeries, according to the WSJ. So far, results have been inconclusive as the children have not reached the age of 5, but the researchers believe that longer exposure or multiple exposures can cause changes in the brains of small children. To that end, they have begun studies using a combination of shorter-term general anesthesia and spinal anesthesia.
"When I tell parents I don't need to place a breathing tube or an anesthesia mask on their baby, the sense of relief on a mother's faces is remarkable," anesthesiologist Robert Williams of the University of Vermont Children's Hospital told the WSJ. He said babies would often be holding his hand during surgery, which was a comfort to the parents. The hospital adopted spinal anesthetics as its first choice for infant surgeries and was one of the technique's pioneers decades ago.
- here's the WSJ piece