 |
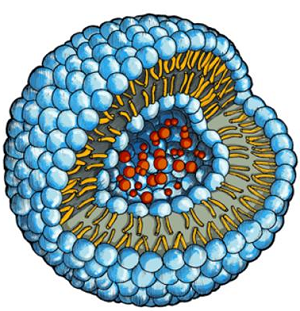
| Drug molecules (red) inside a water-filled cavity of a phospholipid vesicle--Courtesy of U. of Basel |
Researchers in Switzerland have developed a new kind of nanoparticle that is essentially invisible in the human body, an advancement that would allow for drug delivery vehicles that bypass the immune system, particularly in this case to aid stenosed arteries.
Normal liposomes are recognized as foreign bodies, according to a release from the University of Basel, where the researchers conducted their research with help from the University of Fribourg. Because they tend to affect the immune system, they can lead to an allergic reaction with negative effects in up to 30% of cases, which can include anaphylactic shock.
For this study, the scientists used artificial phospholipid vesicles, which did not cause any such reaction, and which could be replicated in live pigs. The particle itself is like a soap bubble made of a double phospholipid membrane with water in its internal cavity, where the drug is also stored.
"The study shows that Pad-PC-Pad liposomes are not inducing direct or indirect anaphylactic reactions, even at high dosages," says Professor Bert Müller of the University of Basel. "These are highly unexpected results (and) could have a high impact of future treatments of atherosclerosis."
As for the promise these nanoparticles hold for coronary artery diseases, as published in the journal Nanomedicine: Nanotechnology, Biology, and Medicine, the drug-carrying vehicles enter the bloodstream, and due to the high velocity therein, they are opened and release their content. With these particles in such a vulnerable area, it is important they go unrecognized by the body's defenses.
- here's the release
- read the journal abstract