 |
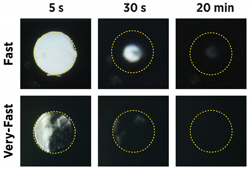
| Two of the UW researchers' fibers (white) are shown dissolving. The material has the potential to release higher levels of antiretroviral drug in less time than topical gels or rings.--Courtesy of University of Washington |
Researchers at the University of Washington have developed a medicated, electrospun fabric that could prevent HIV infections in women.
UW bioengineers spun topical drugs into silklike fibers that quickly dissolve when exposed to moisture, releasing higher levels of antiretroviral drug in less time than topical gels or rings, according to study results published in the August issue of Antimicrobial Agents and Chemotherapy. Researchers hope to create HIV-preventing tampons from the material that could be inserted into the vagina prior to sexual intercourse, offering women a more discreet way to protect themselves from infections, lead study author Cameron Ball said (as quoted by the University of Washington).
"The effectiveness of an anti-HIV topical drug depends partially on high-enough dosages and quick release," Ball said. "We have achieved higher drug loading in our material such that you wouldn't need to insert a large amount of these fibers to deliver enough of the drug to be helpful."
The UW research team created the material through electrospinning, a process that charges a liquid form of drugs with a high-voltage generator and passes the substance through a syringe. The electric charge on the liquid's surface causes it to form a string in the syringe, where it whips around before collecting on a surface. A palm-sized swatch of the material takes 5 minutes to make, and researchers were able to dissolve the drugs in about 6 minutes by adjusting ingredients in the fibers.
UW researchers are not the only ones zeroing in on new delivery methods for HIV-preventing drugs. Last year, scientists at the Washington University School of Medicine in St. Louis developed a nanoparticle gel made from bee venom that, when applied vaginally, could help prevent the spread of HIV. In March, the U.S. National Institutes of Health released mixed Phase I study results for its HIV-preventing vaginal ring. Only one of the ring's two antiretroviral drugs was effective in protecting against the virus, but both drugs proved safe in a 28-day trial.
- read the study abstract
- here's the University of Washington story