 |
| The Viaskin patch, used for food allergies including peanut sensitivity--Courtesy of DBV |
A child's peanut allergy can be a source of constant worry for a parent, and as food sensitivities become more and more common, finding a way to protect children from these allergic reactions could be big business. Over the past few years, France's DBV Technologies ($DBVT) has been financing its food allergy patch, and a new study showing that the patch helps children tolerate more peanuts has boosted the market value to the tune of almost $1 billion, according to a report from Bloomberg.
The Viaskin Peanut patch is set to become the first approved in a therapeutic market bereft of other solutions; for now, avoidance is the only way of preventing allergic reactions. In the past decade, prevalence of severe allergies has risen by almost 2% in children. More than 1% of children in countries involved in studies have a peanut allergy, which is the leading cause of fatal reactions.
In the study, 54% of children showed tolerance for up to four peanuts, more than the amount typically encountered accidentally. The patch itself caused no severe reactions despite some skin irritation.
DBV's patch delivers a small amount of peanut protein to skin cells, which boosts the immune system and allows for better resistance. And that delivery route is thought to be the most convenient for families dealing with a child's daily allergy struggles. The Viaskin platform concentrates allergens in superficial layers of the skin, letting the immune system get a taste without passing the dangerous material into the bloodstream. The company calls this process epicutaneous immunotherapy.
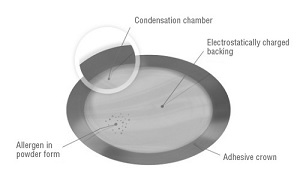 |
| Illustration of the Viaskin patch--Courtesy of DBV |
The patch is undergoing a third round of trials set to begin by early 2016, involving 250 to 400 children, teenagers and adults. The patch is originally made for children, but by increasing the protein dose, the company hopes to achieve the same results for older patients.
Selling shares in the U.S., DBV collected about $100 million in October, which Bloomberg states would allow it to stick to its guns, without the involvement of a big pharmaceutical. They have already turned down a few collaborations for the peanut allergy patch, DBV CFO David Schilansky said, although other partnerships such as last year's $198 million deal with Stallergenes for a birch allergy have paid off.
"It would be a shame to involve a big pharmaceutical company that would pocket two-thirds of the value," Schilansky told the publication. "We have the money, so we can do as we please."
- here's the Bloomberg report