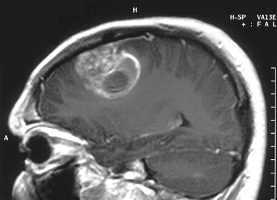
 |
| MRI image of a glioblastoma tumor--Courtesy of NIH |
The quest to find cancer vaccines has been filled with roadblocks, and the story is no different with treatments for glioblastoma--the most common and aggressive type of brain tumor--which has so far eluded efforts to stave it off. But Celldex Therapeutics ($CLDX) has some positive new interim results that show it may one day be able to change that.
In a Phase II trial, its vaccine--rindopepimut, or Rintega--helped recurrent glioblastoma patients who took it with Roche's ($RHHBY) Avastin survive a median of 3.2 months longer than patients who took Avastin alone. While the group that took just Avastin survived for 8.8 months, the addition of Rintega took that to 12 months--a statistically significant benefit.
"The patients in this study have advanced and difficult to treat disease," Dr. David Reardon, clinical director of Dana-Faber's Center for Neuro-Oncology, said in a statement. "While this is interim data, the ability to demonstrate a survival benefit in this patient population has been extremely rare."
 |
| Dr. David Reardon, clinical director of Dana-Farber's Center for Neuro-Oncology |
Celldex will now follow the patients in the study for a further three to 6 months before filing final results. If the final data match the interim results, it would be a game-changer for physicians trying to treat glioblastoma and a source of "new hope" for patients and their families, Reardon said.
Cancer vaccine makers have so far faced a host of challenges, with companies like GlaxoSmithKline ($GSK) and Merck KGaA recently announcing late-stage failures that cast doubts on the field. In glioblastoma specifically, Northwest Biotherapeutics ($NWBO) has touted individual success stories from patients in a Phase I/II study of prospect DCVax-L who lived beyond the expected survival time. But critics have said the data shouldn't be considered statistically significant because of the trial's size and "informal nature."
- read the release
Special Report: ASCO preview: What's the outlook for cancer vaccines?