 |
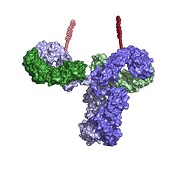
| Site-specific antibody drug conjugates on a molecule--Courtesy of Ambrx |
San Diego-based Ambrx welcomed an influx of cash for the further development of its drug-delivery technology: antibody drug conjugates, or ADCs.
Japanese drugmaker Astellas Pharma will pay $15 million up front to license the company's technology and another $285 million as Ambrx reaches milestones in research, development, regulation and sales.
Ambrx, a FierceBiotech 2005 Fierce 15 company, designs its ADCs to mimic the antibodies in the human body in a way that allows drugs to more efficiently and safely reach their targets, according to the company. In cancer treatments, for instance, ADCs help the toxic drugs to reach tumors based on specific receptors on the surface of the offending cells.
Ambrx's lead drug candidate is ARX201 for growth deficiency, which has completed Phase II trials. Astellas has partnered with the company specifically for cancer.
Antibodies as a drug-delivery tech are gathering a lot of interest in the pharmaceutical arena. Roche's ($RHHBY) Genentech picked up FDA approval for its ADC-powered breast cancer therapy Kadcyla, formerly T-DM1, in February. Ambrx notes several advantages of its ADCs over conventional conjugation treatments, however, including compatibility with more drugs, a tighter link with those drugs and minimized unintended effects.
And Astellas is only the latest to latch onto Ambrx's version of ADC technology, as Merck ($MRK), Eli Lilly ($LLY) and Bristol-Myers Squibb ($BMY), among others, have Ambrx-partnered drugs in the pipeline, as well.
- here's the release
Special Report: Ambrx - 2005 Fierce 15 Revisited