Recalls for Baxter ($BAX) continue to pile up, with the drugmaker retrieving an injectable product this time because of particulate. The recall comes just weeks after it had to retrieve another injectable because of labeling problems.
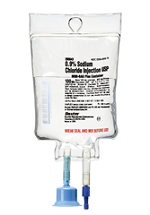
The Deerfield, IL, company said Tuesday that it was voluntarily recalling from hospitals and users two lots of 0.9% sodium chloride injection in what it calls 100 mL MINI-BAG Plus Container, after getting two complaints, one per lot, that a bag contained particulate. In this case, the company said the particulate turned out to be "frangible from the vial adapter." There have been no complaints of adverse reactions but the notice distributed by the FDA carried the usual caution that injecting particulate can lead to a blockage that could cause a stroke or heart attack.
While there has been a national shortage of 0.9% sodium chloride injection, or saline, in large containers that hospitals typically use, the company in an emailed statement said there is no shortage of 0.9% sodium chloride injection "in the 100 mL MINI-BAG Plus Container, and the recall will not create a disruption to patient care. Unaffected lot numbers can continue to be ordered."
Baxter has had a steady stream of recalls this year for a variety of reasons from mold in a dialysis product to mislabeled containers. Several weeks ago, Baxter voluntarily recalled one lot of highly concentrated potassium chloride after a complaint the overpouch of some product was labeled as having twice the dose. The problem, as Baxter pointed out in its recall, is that patients may not be given the right dose, a particular danger for patients in the high-risk category who are "prone to severe electrolyte imbalance." In September, one lot of potassium chloride had to be taken off the market because some shipping cartons were discovered to contain units of the antibacterial drug gentamicin sulfate injection.
In August, Baxter recalled a single lot of 0.9% sodium chloride injection, in 1000-mL bags, after particulate matter was found near the administration port in some containers. It is the dosage most affected by a national shortage that has plagued hospitals this year. In July, the drugmaker recalled two lots of saline and another product from Chile, Hong Kong, Singapore and the U.S. A spokesman said at the time that the recall of sodium chloride was smaller volume products that would not be meaningful to a saline shortage in the U.S.
- here's the recall notice