 |
| Courtesy of NIH |
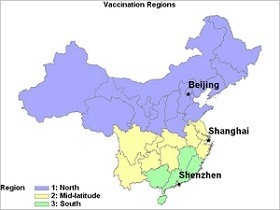
China is a big, climatologically-diverse country, with the weather changing significantly between the temperate zones in the north and the subtropical regions further south. This affects when flu seasons occur, and consequently when health authorities should run vaccination campaigns.
A team of U.S. and Chinese researchers have looked into this topic aiming to help the country plan a data-driven vaccination strategy. The researchers, who included National Institutes of Health staffers, found that Beijing and other cities in the northern band of China are similar to temperate zones around the world. Flu season peaks over the winter, so vaccination campaigns are best run around October. In the south of the country, around Shenzhen, the subtropical climate means flu season peaks in the spring.
The difference suggests a staggered vaccination campaign would be most effective, with the southern zone offering vaccines from February onward. Shanghai and other mid-latitude cities complicate the picture, though. Over the 7-year period analyzed by the researchers, the mid-latitude region experienced semi-annual flu peaks in the winter and summer. The data suggest the region should be classed separately from the southernmost area, but further research is needed to plan an optimal vaccination strategy.
With China offering such a huge opportunity for vaccine manufacturers, the outcome could affect supply plans. GlaxoSmithKline ($GSK), Sanofi ($SNY) and other vaccine manufacturers, both local and global, have all established a presence in China's seasonal flu shot market, but so far uptake is limited. Around 2% of people receive the vaccine, but private use is expected to increase as the country becomes wealthier. And if government subsidy programs expand, China could become a major market for flu vaccine players.
- read the NIH release
- here's the paper