 |
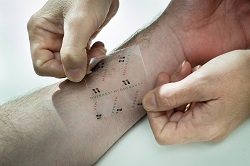
| Medherant's transdermal patch--Courtesy of University of Warwick |
University of Warwick spinout Medherant has a developed a transparent adhesive patch that can deliver the painkiller ibuprofen transdermally for up to 12 hours. It hopes the breakthrough paves the way for the development of additional long-acting over-the-counter therapies for conditions like chronic back pain, neuralgia and arthritis.
According a university news release, the novel patch can remain adherent to the skin with drug loads 5 to 10 times greater than used in current medical patches and gels. It is easy to remove.
The patch deploys new polymer technology developed by global adhesives company Bostik that was exclusively licensed to Medherant for transdermal use.
"Many commercial patches surprisingly don't contain any pain relief agents at all, they simply soothe the body by a warming effect. Our technology now means that we can for the first time produce patches that contain effective doses of active ingredients such as ibuprofen for which no patches currently exist. Also, we can improve the drug loading and stickiness of patches containing other active ingredients to improve patient comfort and outcome," said University of Warwick chemistry professor David Haddleton in a statement.
He added that patch has achieved "great results" in tests of methyl salicylate delivery, which is used in some leading commercial patches.
Meanwhile, Medherant CEO Nigel Davis said that through partnering, the company hopes have its first product on the market in two years. "Our transdermal patch technology expands the range of drugs that can be delivered via skin patches and can significantly increase drug loading capabilities, whilst retaining adhesion and being thin and flexible. Thus our patches provide a better experience for patients, enhance safety and deliver increased efficacy, which will lead to economic benefits to the healthcare system," he said.
- read the release