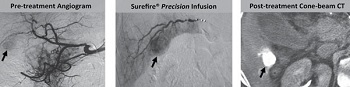
 Surefire Medical picked up FDA clearance for its embolization device designed to deliver cancer drugs directly to tumors. The 510(k) comes on the heels of clinical data published in the Journal of Vascular and Interventional Radiology.
Surefire Medical picked up FDA clearance for its embolization device designed to deliver cancer drugs directly to tumors. The 510(k) comes on the heels of clinical data published in the Journal of Vascular and Interventional Radiology.
Surefire's Precision tool delivers cancer drugs with an extendable tip. The device allowed increased uptake of particles by an average of 68%, according to the study, and as high as 90% in some cases compared to common catheters.
"This is a promising tool that may help to improve clinical outcomes in patients undergoing liver directed intra-arterial therapies for primary and secondary liver cancers," Georgetown University Medical Center's Alexander Kim said in a statement.
Surefire picked up $11 million in June to back its microcatheter system Infusion.
"We developed this new drug delivery device for treating the large worldwide primary liver cancer population, and potentially several other clinical applications" said James Chomas, CEO of Surefire Medical. "Whether as a first-line treatment, palliative care, or for bridging patients to a liver transplant, the technologically advanced Surefire Precision enables physicians to improve targeting in minimally invasive, direct-to-tumor cancer treatments."
- here's the release