 |
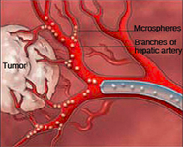
| A depiction of "embolotherapy," or the delivery of drug-loaded microspheres to the site of a tumor using a catheter (blue)--Courtesy of EmboMedics |
Devicemaker AngioDynamics threw its hat into the cancer drug delivery ring through its partnership with EmboMedics, the developer of investigational chemotherapy-delivering microspheres that also perform embolization.
The deal calls for AngioDynamics to receive an exclusive license to market and sell the drug delivery technology upon FDA clearance. In return, the company will make an initial $2 million equity investment in EmboMedics, which could rise by an additional $9 million based on milestones. If all goes well, AngioDynamics can execute its exclusive option to buy the partner, according to a release.
Embolization involves blocking blood flow to arteries by introducing foreign materials near the target using a catheter. The method, called "embolotherapy," is used to treat a variety of conditions including aneurysms, bleeding and cancer.
EmboMedics is studying and developing microspheres that both deliver drugs and block blood flow to cancerous tumors. It is using a new bioresorbable bead technology, with hopes of submitting the candidate for FDA clearance as a medical device to treat hypervascular tumors by 2016. That is significant because FDA sometimes requests that device/drug combination products be regulated via the drug pathway. Such a decision would significantly lengthen the time to approval.
"We believe resorbable microspheres represent the future of embolotherapy, allowing for more options in the treatment of patients, and we are excited about the prospect of bringing this technology to our customers worldwide," said AngioDynamics CEO Joseph DeVivo in a statement.
 |
| AngioDynamics CEO Joseph DeVivo |
EmboMedics CEO Dr. Omid Souresrafil said his company will benefit from AngioDynamics "world class sales force and distribution network." That's because AngioDynamics makes a variety of complimentary products, including catheters and interventional devices for oncology, such as the NanoKnife for surgical ablation (or removal) of soft tissue.
The move comes as AngioDynamics looks to reorient itself following a disappointing quarter that featured a 2% reduction in year-over-year sales to $86.6 million. During the earnings call, the company announced that it will permanently yank its drug delivering Morpheus peripherally inserted central catheter from the market.
- read the release